










 GENTAUR BVBA
GENTAUR BVBA
VAT BE0473327336
Av. de l Armee 68 B4
1040 Brussels
BELGIUM
Tel + 32 16 58 90
45
Fax + 32 16 50 90 45
 GENTAUR France SARL
GENTAUR France SARL
SIRET 48423788800017
Rue Lagrange, 9
75005 Paris,
France
Tel 01 43 25 01 50
Fax 01 43 25 01 60
 GENTAUR Germany
GENTAUR Germany
52074 Aachen,
Germany
Tel +32
16 58 90 45
Fax +32 16 50
90 45
 GENTAUR
Pol Sp. Z.o.o. Ulica
Ogarna 15/19B m2 GENTAUR
Pol Sp. Z.o.o. Ulica
Ogarna 15/19B m2
80-826 GDANSK
Tel 00 48 51 760 77
08
Fax: 00 32 16 50 90
45
 GENTAUR Italy
GENTAUR Italy
23015 Milano, Italy
Tel 02 36 00 65
93
Fax 02 36 00 65
94
 Česká republika
Praha
Česká republika
Praha
+420246019719
 Danmark
Danmark
+4569918806
 Finland Helsset
Finland Helsset
+358942419041
 Ελλάς Αθήνα
Ελλάς Αθήνα
+302111768494
 Ireland Dublin
Ireland Dublin
+35316526556
 Luxembourg
Luxembourg
+35220880274
 Magyarország
Budapest
Magyarország
Budapest
+3619980547
 Nederland
Nederland
+31208080893
 Norge
Oslo Norge
Oslo
+4721031366
 Österreich
Österreich
+43720880899
 Sverige
Stockholm Sverige
Stockholm
+46852503438
 Schweiz Züri
Schweiz Züri
+41435006251
Northern America
 Canada Montreal
Canada Montreal
+15149077481
 US New York
US New York
+17185132983
Other Countries

0032 (0)16 41 44 07 |
|
|
|
|
|
|

| |
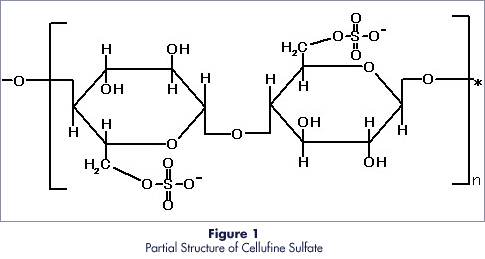
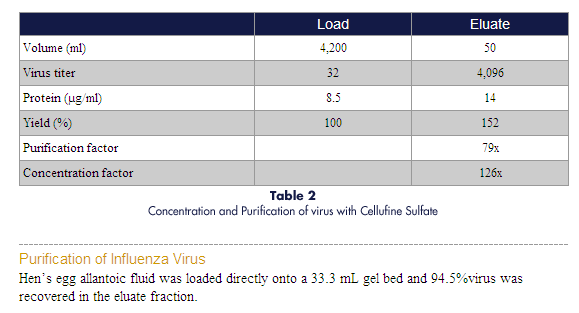
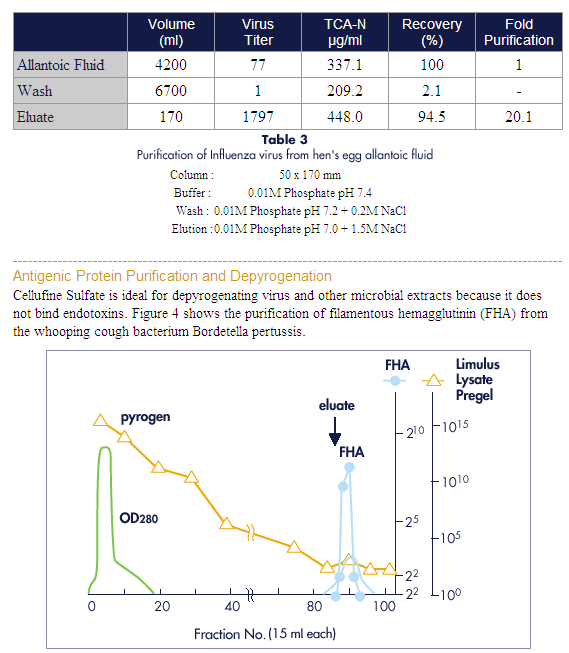
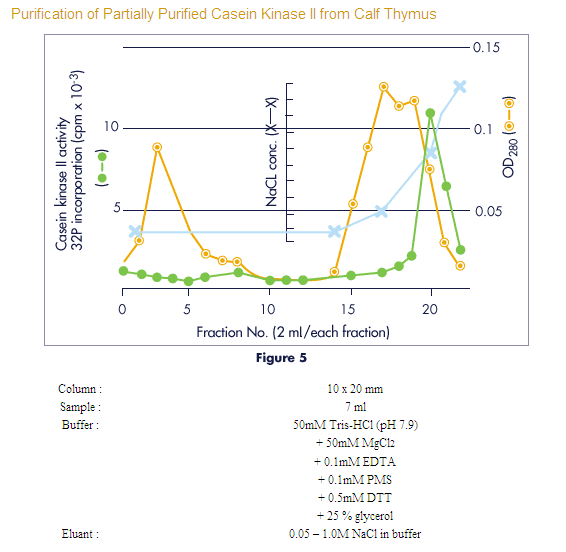
Cellufine Sulfate
For concentration, purification and depyrogenation of virus, viral/microbial
antigens and heparin-binding proteins.
Advances in vaccines and clinical diagnostics have created an increasing
demand for large volumes of highly purified and concentrated virus and viral or
microbial antigens. Cellufine Sulfate affinity media is a simple, rapid and
effective means for concentration, purification and depyrogenation of these
important products.
Cellufine Sulfate eliminates cumbersome, time-consuming and potentially
unsafe classical ultra-centrifugation and density gradient methods. It can also
provide a significant improvement in concentration and purity. Cellufine Sulfate
can reduce or eliminate the expense, ligand leakage and reproducibility problems
associated with immobilized dextran sulfate, chondroitin sulfate or heparin.
Elution of the bound product is affected through simple stepwise or gradient
increases in ionic strength.
Features:
• Affinity for a wide range of live, killed or disrupted viruses, viral or
microbial antigens and heparin-binding proteins
• Closed column operation assures safety and product sterility
• Endotoxins do not bind, allowing a rapid and contaminant free depyrogenation
• Rigid, high-strength beads
• Autoclavable
Benefits:
• More effective than ultracentrifugation at removing contaminants from culture
media and host cells
• Avoids excessive product handling and safety concerns, particularly with viral
preparations
• Simultaneous concentration and purification improve yield, reduce processing
steps, time and costs
• Gentle binding and elution conditions provide high capacity and product yield
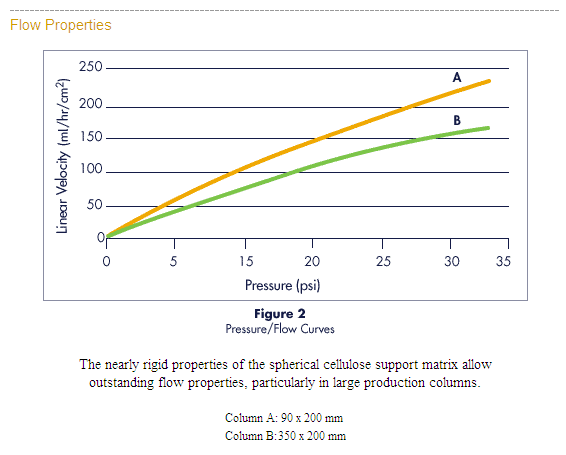
• Resists compression, providing rapid flow for high-speed processing, even in
large columns, making it easily scalable
• Resistant to chemical depyrogenation with base and chemically sterilizable
with formalin


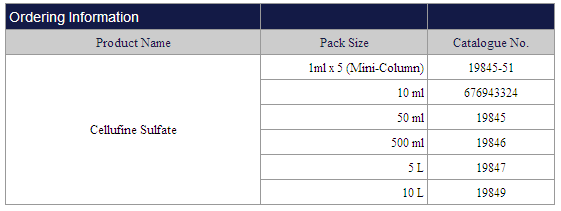
|
![]()
![]()
![]()
![]()
![]()
